Instructions For After The Procedure
You will have follow up appointments with your doctor to make sure the device is programmed in the best way to control your symptoms. You will receive a remote control device to control stimulation, and you may see the InterStim nurse or technician as needed to adjust settings for optimal benefit. This may take a few visits after the initial implant. Once the most appropriate settings are determined, you will need to be followed annually or as needed. Patients can expect up to 6 years of benefit before the device may need to be replaced due to battery depletion.
What Is Interstim Micro
The U.S. Food and Drug Administration recently approved the InterStim Micro neurostimulator, the markets smallest and fastest rechargeable device to deliver sacral neuromodulation therapy. It offers a smaller size compared to InterStim II and a longer battery life. InterStim Micro also allows full-body 1.5 and 3 Tesla MRI conditional scans with SureScanTM MRI technology.
How The Interstim Ii System Works
Step 1: The Interstim II implant lead is placed alongside the sacral nerve in the lower back.
Step 2: The removable implant transmits a signal from the bladder to the brain to indicated fullness of the bladder.
Step 3: The Interstim II System treatment provides reliable bladder control by restoring effective communication between the brain and bladder.
Recommended Reading: How To Take Azo Urinary Tract Defense
What Is Interstim Ii
The recharge-free InterStim II system gives patients freedom from a recharging routine, the hassle of recharging components, and a reminder they have a disease. InterStim II is simple, convenient, low maintenance. InterStim II now allows full-body 1.5 and 3 Tesla MRI conditional scans with SureScanTM MRI technology.
The Relief Youve Been Waiting For
- Targets the nerves that control your bladder to help it function normally again
- 85 percent of people with OAB using it achieved success in the first year4**
- 3X greater improvements in OAB quality of life compared to medications5
- Only therapy that lets you see if it works before you and your doctor decide
- More than 350,000 patients have received relief as a safe, FDA-approved and minimally invasive Bladder Control Therapy
- Recharge-free and rechargeable options let you choose the right device for your lifestyle
In addition to risks related to surgery, complications can include pain at the implant sites, new pain, infection, lead movement/migration, device problems, undesirable changes in urinary or bowel function, and uncomfortable stimulation . Talk with your doctor about ways to minimize these risks.
Quarter shown for size comparison
You May Like: Azithromycin For Urinary Tract Infection
Read Also: Urinary Tract Infection Antibiotics Walmart
Risks Of Interstim Therapy
As with any minimally invasive procedure, patients have a chance of infection, swelling, bruising and bleeding. Pain medication will be prescribed to help with pain and discomfort.
Patients with an InterStim Therapy device will not be able to have an MRI or be subjected to other kinds of diagnostic equipment. If a patient becomes pregnant or is trying to become pregnant, the doctor will need to shut off the device.
Request an Appointment
Who Is A Candidate For Treatment With Sacral Neuromodulation
InterStim® therapy is approved for usage by Medicare in Australia in people older than 18 years of age whose symptoms have not responded to medical and conservative treatments over at least 12 months due to:
- Overactive bladder due to detrusor overactivity or
- Paradoxically also in patients with urinary retention which is not due to a blockage in the urinary tract
In Australia InterStim® therapy is also approved for use in some patients with refractory faecal incontinence which has not responded to other treatments.
Don’t Miss: Tea Tree Oil Urinary Tract Infection
What Is Involved In The Permanent Pacemaker Or Stage 2 Interstim Implant
The patient and doctor decide together before the 2nd stage procedure if the InterStim® device has made enough of a difference to symptoms to proceed to the permanent pacemaker implant.
If this is the case, under an anaesthetic a small cut is made in the buttock skin and the pacing wire is connected to the permanent InterStim® pacemaker device which is implanted deep to the fat of the buttock.
The device is controlled by a hand held patient programmer with no external visible wires. Patients are given extensive education in how to use the patient programmer that is usually no more complicated to use than a mobile phone. Usually settings do not need to be changed on a regular basis using the patient programmer.
The InterStim® device is left on continuously and fine-tuning of programmes or settings occurs over the first few months depending on urinary symptoms.
What Are The Benefits Of The New Interstim Systems For Patients With Oab Or Fi
Medtronic is the only company to offer patients the freedom to choose between a rechargeable or recharge-free sacral neuromodulation device to best match their preferences, lifestyle and treatment goals. Both InterStim Micro and InterStim II are full body MRI conditional, and deliver the same therapy and long-term relief.
The recharge-free InterStim II system is simple and convenient with lower maintenance and time commitments. This specific system gives patients the freedom from a recharging routine, the hassle of recharging components, and a reminder they have a disease.
The InterStim Micro rechargeable system is the smallest device on the market with the fastest rechargeability and is stronger than other manufacturers batteries. It features proprietary Overdrive battery technology a battery with virtually no loss in capacity over time. The new battery technology allows patients to choose how and when they want to charge their device from as often as once a week, or as infrequent as once per month, depending on the patients preference and device settings. There is no battery fade at 15 years* and patients can restart their therapy after extended breaks in time.
To help decide which system best fits your needs and lifestyle, please speak with our providers and watch the video below.
Read Also: How Does Kidney Failure Affect The Urinary System
Lasting Relief At Last
Medtronic bladder control therapy delivered by the InterStim II system provides life changing relief. And the InterStim Micro system is designed to deliver the same eective results.
- 84% satisfaction among those who use it7,¶
- 76% of people achieved success at 6 months compared to 49% who used medications4,#
- The only OAB therapy that provides better relief than medications4,8
The most common adverse events experienced during clinical studies included pain at implant sites, new pain, lead migration, infection, technical or device problems, adverse change in bowel or voiding function, and undesirable stimulation or sensations. Any of these may require additional surgery or cause return of symptoms.
Interstim Bladder Control Therapy
If youre struggling with urinary control and the symptoms of overactive bladder, Medtronic Bladder Control Therapy may be able to help you.
Medtronic Bladder Control Therapy, also known as InterStim Therapy for Urinary Control, is used for the treatment of urinary retention and the symptoms of overactive bladder including urinary urge incontinence and significant symptoms of urgency-frequency. Generally, it is used for patients who have not responded to more conservative treatment options.
Recommended Reading: Difference Between A Urinary Tract Infection And A Bladder Infection
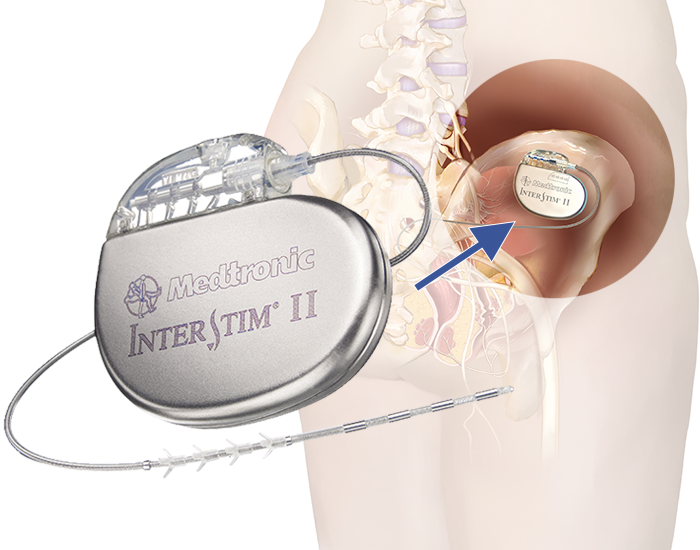
What Is The Interstim Ii System
The InterStim II System is a medical-grade implant designed to deliver controlled electrical stimulation to the sacral nerve. This is believed to restore normal communication between the brain and bladder. This sacral neuromodulation system has been proven safe and effective to restore function, significantly improve quality of life for OAB patients, and provide long-term control
How Is Interstim Implanted
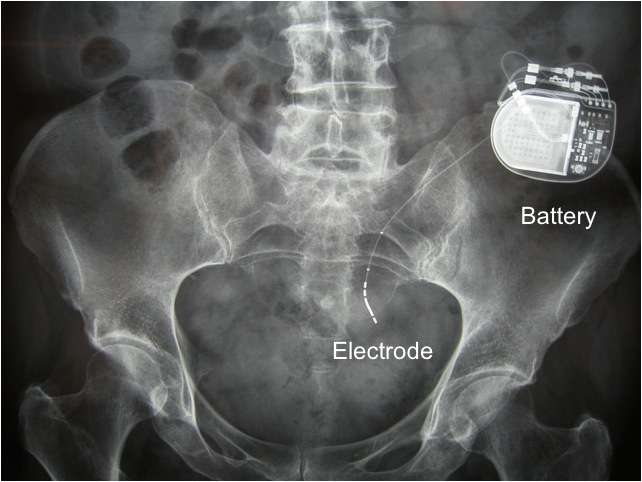
The device is actually not implanted anywhere near the bladder. It is placed in the lower back area, near the nerves that control bladder function.
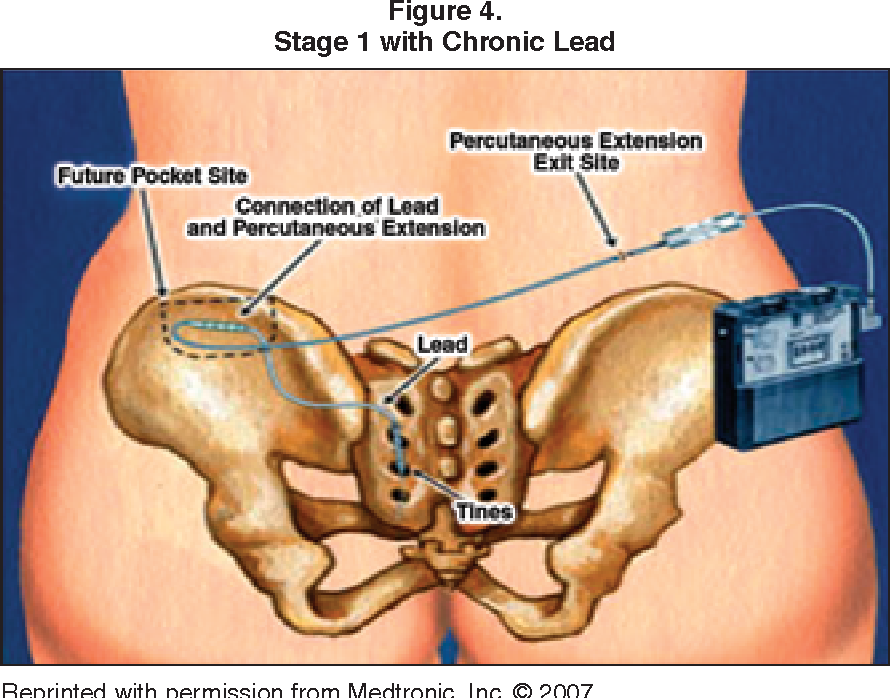
Inserting the Interstim device is a 2-step process:
Phase 1: A lead is placed in the lower back, under mild sedation in the operating room. A couple of small incisions are used to implant the lead, and then we do a test run for a week or two to see how it affects your symptoms. If the test is successful, and you notice at least 50 % improvement in your number of trips to the bathroom and/or bladder accidents, then we proceed to the second phase.
Phase 2: The intermittent pulse generator is implanted. This very simple procedure creates about a half-inch pocket under the skin in the buttock area. Then, the lead is plugged into the generator and absorbable stitches close the wound.
Read Also: What Not To Eat With A Urinary Tract Infection
Interstim Therapy For Urinary Incontinence
Urinary incontinence affects over 25 million Americans. A number of causes exist: injury or trauma to the pelvic region or back, enlarged prostate or prostate cancer, pregnancy or childbirth, neurological disorders, estrogen loss following menopause, other health conditions like multiple sclerosis or suffering a stroke.
Take our Urinary Incontinence Self-Assessment
How The Procedure Is Performed
Unlike other bladder control treatments, InterStim lets you try it first with an evaluation. The evaluation is performed right in your doctors office and involves placing a lead in the upper part of your buttock. The lead then attaches to a small external device worn discreetly under your clothes.
Most patients can tell within just a few days if its working for them, though some evaluations may take up to two weeks. After the evaluation period, you and your doctor will decide if the long-term therapy is right for you. If it is, your evaluation device will be replaced with an implantable device called a neurostimulator during a short outpatient procedure.
Recommended Reading: Hills Urinary Care C D Multicare
What Is Medtronic Bladder Control Therapy
Medtronic Bladder Control Therapy , delivered by the InterStim System, uses an implantable device that sends gentle electrical stimulation to the sacral nerves, located near the tailbone. The sacral nerves control the bladder and the muscles related to urination. If the brain and sacral nerves dont communicate correctly, the nerves cant tell the bladder to function properly. The gentle electrical stimulation from the InterStim System corrects this miscommunication.
Medtronic Bladder Control Therapy is an option for managing symptoms of urinary retention for people who have not found success with more conservative treatments, and whose urinary retention is not caused by a blockage such as cancer or an enlarged prostate.
Medtronic Bladder Control Therapy is reversible and can be discontinued at any time by turning off or removing the device.
Medtronic Bladder Control Therapy may help you resume normal activities, and help you avoid frustrating experiences associated with urinary retention.
Benefits Of Interstim Therapy
InterStim therapy is a simple, outpatient surgical procedure that is shown to give life-changing and long-lasting relief for symptoms of overactive bladder. InterStim therapy can be tried externally before receiving the implanted device. Your doctor can place a temporary external nerve stimulating device over the sacral nerves in the upper buttocks to perform a peripheral nerve evaluation and determine if this procedure is right for you. Over the course of a few days, your symptoms may improve by 50% or more, which indicates you are a candidate for InterStim therapy.
Also Check: What Does A Urinary Tract Infection Feel Like
Medtronic Bladder Control Therapy Delivered By The Nuro System
- Targets the tibial nerve to help you regain control of your bladder
- Does not cause unpleasant side eects like many oral medications can10
- Does not require self-catheterization, unlike injectable medications8
- Helps you live with less worry and more condence
The NURO system only treats the symptoms of OAB, not retention.
Most common side eects of PTNM are temporary and include mild pain or skin inammation at or near the stimulation site.
Effectiveness Of Interstim Devices For Bowel Incontinence
In study results published in the medical journal Annals of Surgery, 120 patients and their doctors tracked the effectiveness of the therapy.
- About half of the patients experienced total recovery of bowel control and reported no incontinence problems for one year after the surgery
- 30% reported that their bowel leaks were reduced by more than 50%
- InterStim therapy produced significant positive results for more than 8 out of 10 patients
Recommended Reading: Back Pain Causing Urinary Problems
How Interstim Therapy Works
Your sacral nerves control and regulate your bladder. If theyre damaged, this can lead to incontinence, because the signals between your brain and your bladder can get mixed up. If your brain receives a signal at the wrong time, it can lead to an unneeded urge to urinate. The FDA-approved InterStim system aims to improve communication along the sacral nerves by introducing electrical stimulation.
Nerve damage can result from a number of underlying health conditions, such as diabetes, a nervous system disorder, a spinal injury, an infection, or childbirth.
What To Expect During The Procedure
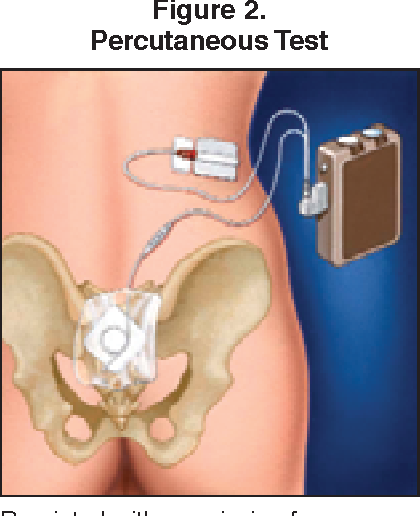
Implanting the InterStim neurostimulator is a minimally invasive surgical procedure. Your board-certified urologist will insert the lead near your sacral nerves. The other end of the lead will be connected to the neurostimulator. You will be able to control the neurostimulator with an external device that can adjust the stimulation or turn the device off.
Read Also: Menâs Urinary Tract Problems
Recommended Reading: How Does A Urinary Tract Infection
Who Is A Candidate For Interstim
Interstim is designed for women with an overactive bladder who go to the bathroom too often, suddenly lose urine because of a bladder spasm, or cannot make it to the bathroom in time. Many of these women have tried numerous medications and pelvic floor exercises, that have failed, and they have what we call refractory overactive bladder or refractory urge incontinence.
If it seems like nothing is working for your overactive bladder, Interstim may finally solve your problem.
Medtronic Interstim Therapy Evaluation
If you’ve tried several treatments for bladder control problems without success, don’t lose hope. Medtronic InterStim Therapy may be an option for you.
With InterStim Therapy’s two-step process, you can test it out to see if it will work for you before making a long-term commitment. The testing period is called an “evaluation” or a “trial assessment.”
Your doctor will discuss the evaluation procedure with you and the options for using either a temporary lead or long-term lead for the evaluation.
Read Also: What Are The Symptoms Of Urinary Tract Infection In Males
How Long Has Sacral Neuromodulation Been Around
The InterStim System received its CE mark for the treatment of chronic intractable function disorders of the pelvis, lower urinary and intestinal tract in 1994. Today over 225,000 patient have been treated worldwide. Itâs a proven treatment option for people who have not had success with more conservative urinary retention treatments.
What Is Interstim Therapy For Urinary Control
Medtronic InterStim® System for Urinary Control is indicated for the treatment of overactive bladder/urge incontinence, non-obstructive urinary retention and fecal incontinence, in patients who have failed or could not tolerate more conservative treatments.
InterStim Therapy uses a small device to send mild electrical pulses to a nerve located in the lower back This nerve, called the sacral nerve, influences the bladder, bowel, and surrounding muscles that manage urinary and bowel function. The electrical stimulation may eliminate or reduce certain bladder and bowel control symptoms in some people.
The sacral nerves, located near the sacrum , play an important role in regulating bowel and bladder control. A sacral nerve neurostimulator, known as InterStim, may help some patients with refractory urge incontinence, as well as individuals with non-obstructive voiding dysfunction and those with fecal incontinence. The system uses a small implantable device to send electrical pulses to the sacral nerves. OAB and Urge Incontinence are caused by abnormal bladder contractions and the Interstim neuromodulator interrupts these abnormal bladder contractions, thus controlling incontinence. The InterStim system is currently FDA approved for:
- Refractory Urge Incontinence
- Non-obstructive voiding dysfunction
- Fecal Incontinence
Read Also: Royal Canin Urinary Tract Food
Stage : The Implantation Of The Internal Stimulator
This procedure is performed under local anesthesia with sedation
- The incision on the buttock area from the first surgery is reopened and a pocket is made in the fatty tissue and the generator placed inside Interstim final implant
- The outside wire is removed
- The procedure takes about 30 minutes and you can go home the same day.
- You should again limit for activities for 7-10 days.
Learn How Interstim Can Remedy Your Urinary Incontinence
Depending on the severity and underlying cause of your incontinence or bladder control issues, conservative treatments, such as medication and behavior modification, may not be enough.
If you struggle to manage your symptoms effectively, you may be a good candidate for InterStim, a type of neuromodulation therapy.
Ahmad Kasraeian, MD, and Ali Kasraeian, MD, offer neuromodulation therapy for urinary incontinence at Kasraeian Urology. Neuromodulation therapy is an advanced treatment that can improve how your brain communicates with the nerves that control your bladder function.
Don’t Miss: Lower Back Pain Causing Urinary Problems
The Process For Getting An Implant
The InterStim system consists of a neurostimulator and an electrode both of which are implanted in the lower back with small incisions and a wireless remote control. The neurostimulator sends the signals to the electrode, and the electrode stimulates the sacral nerves. The external wireless device allows you to adjust the signal level and turn the system on and off.
Youll be placed under anesthesia for your procedure, which is performed on an outpatient basis and typically takes about an hour. Unlike other surgical treatments, you can take InterStim for a test run to see if its right for you before making a long-term commitment. The initial evaluation period spans 3-14 days, in which time you can go about your normal routine and track your symptoms to see if there are noticeable improvements in your urge, control and frequency.
What Are The Benefits And Potential Side Effects Or Complications Of Medtronic Bladder Control Therapy
Medtronic Bladder Control Therapy may reduce symptoms for many people who suffer from urinary retention. It can also significantly improve quality of life. Medtronic Bladder Control Therapy is reversible and can be discontinued at any time by turning off or removing the device. Unlike other surgical treatment options for urinary retention, Medtronic Bladder Control Therapy starts with an evaluation. During this evaluation you can experience how it feels and how it works, prior to going forward with the implant procedure.
Implanting the InterStim System has risks similar to any surgical procedure, including swelling, bruising, bleeding, and infection. Talk with your doctor about ways to minimise these risks.
Medtronic Bladder Control Therapy might cause you to experience some of these side effects:
- Pain at the implant site or new pain
- Infection or skin irritation
- Interactions with certain other devices or diagnostic equipment
- Undesirable changes in urinary or bowel function
- Uncomfortable stimulation
Problems may be resolved with surgery, medication, or programming. These events may also resolve over time. There is a possibility that some may remain unresolved. See Important Safety Information for possible adverse events. Please consult your doctor. This therapy is not for everyone. A prescription is required.
Recommended Reading: Urinary Tract Infection After Period
Read Also: Hills Urinary Care C D